Tässä artikkelissa sukelletaan syvälle neuroplastisuuteen ja sen syvälliseen vaikutukseen kognitiiviseen kasvuun. Artikkeli tarjoaa toimivia tapoja parantaa neuroplastisuutta ja aivojen sopeutumiskykyä. Tutustu kognitiivisiin harjoituksiin, mindfulnessiin ja aistien käyttöön aivojen joustavuuden edistämiseksi. Tutustu liikunnan, ravinnon, unen ja optimaalisen neuroplastisuuden välisiin yhteyksiin.
Johdanto
Neuroplastisuus, joka tunnetaan myös nimellä aivojen plastisuus tai neuraalinen plastisuus, tarkoittaa aivojen kykyä järjestää itsensä uudelleen muodostamalla uusia hermoyhteyksiä ja muuttamalla olemassa olevia. Sitä voidaan kutsua myös prosessiksi, johon liittyy aivojen mukautuvia rakenteellisia ja toiminnallisia muutoksia. Neuroplastisuus on muuttanut käsitystämme aivoista ja tarjonnut tieteellisen perustan ihmisaivojen huomattavalle joustavuudelle ja sopeutumiskyvylle(1) .
Nykyaikaisen neurotieteen isä Santiago Ramon y Cajal esitti neuroplastisuuden idean ensimmäisen kerran 1900-luvun alussa(2) . Käsite sai kuitenkin laajaa tunnustusta vasta 1900-luvun jälkipuoliskolla neurotieteellisen tutkimuksen ja kuvantamisteknologian kehityksen ansiosta.
Tutkimukset ovat osoittaneet, että aivot muuttuvat jatkuvasti vastauksena sisäisiin ja ulkoisiin ärsykkeisiin(3) . Jokainen kokemus, ajatus ja tunne voi muuttaa aivojemme rakennetta ja toimintaa. Esimerkiksi uuden taidon oppiminen, kuten soittimen soittaminen, jongleeraaminen tai uuden kielen puhuminen, voi johtaa uusiin hermosolujen välisiin yhteyksiin. Samaan aikaan traumaattiset tapahtumat voivat johtaa yhteyksien katoamiseen.
Neuroplastisuuden taustalla oleviin mekanismeihin liittyy muutoksia hermosolujen välisten yhteyksien voimakkuudessa ja määrässä sekä uusien hermosolujen ja synapsien muodostumista. Näitä muutoksia ohjaa geneettisten, epigeneettisten ja ympäristötekijöiden, kuten liikunnan, ruokavalion, stressin ja sosiaalisen vuorovaikutuksen, monimutkainen vuorovaikutus(4) .
Yksi neuroplastisuuden jännittävimmistä vaikutuksista on se, että sitä voidaan hyödyntää toipumisen ja kuntoutumisen edistämiseksi vamman tai sairauden jälkeen. Esimerkiksi aivohalvauspotilailla intensiivinen kuntoutus voi edistää uusien yhteyksien kasvua aivoissa ja parantaa motorisia toimintoja. Vastaavasti mindfulness-meditaatio voi vähentää kipusignaaleja käsittelevien aivoalueiden määrää kroonisesta kivusta kärsivillä henkilöillä.
Neuroplastisuuden eri tyypit
Neuroplastisuus voidaan jakaa karkeasti kahteen päämekanismiin: rakenteellinen plastisuus ja toiminnallinen plastisuus. Yksilön aikajanan osalta neuroplastisuus voidaan myös jakaa kahteen vaiheeseen - kehitysplastisuus ja aikuisen plastisuus. Nämä ovat kaksi neuroplastisuuden näkökohtaa, jotka ilmenevät eri elämänvaiheissa.

Rakenteellinen plastisuus
Rakenteellinen plastisuus viittaa aivojen fyysisiin muutoksiin, kuten synapsien muodostumiseen tai häviämiseen, dendriittisten selkärankojen kasvuun tai vetäytymiseen ja neuronien syntymiseen tai häviämiseen. Näiden muutosten uskotaan olevan taustalla aivojen kyvyssä sopeutua uusiin ympäristöihin ja kokemuksiin, ja ne ovat erityisen tärkeitä kehityksen aikana, jolloin aivot kasvavat ja muuttuvat nopeasti(5).
Toiminnallinen plastisuus
Toiminnallinen plastisuuspuolestaan tarkoittaa hermopiirien toiminnallisten ominaisuuksien muutoksia, kuten synaptisten yhteyksien voimakkuuden muutoksia tai muutoksia hermoverkkojen toimintamalleissa. Nämä muutokset ovat perustana aivojen kyvylle oppia ja muistaa sekä sopeutua muuttuviin kognitiivisiin vaatimuksiin ja ympäristöolosuhteisiin.(6)
Rakenteellinen ja toiminnallinen plastisuus ovat usein riippuvaisia toisistaan, ja muutokset toisessa mekanismissa vaikuttavat toiseen. Sekä rakenteellinen että toiminnallinen plastisuus ovat neuroplastisuuden kriittisiä osatekijöitä, joiden ansiosta aivot pystyvät sopeutumaan ja muuttumaan kokemusten ja ympäristöärsykkeiden vaikutuksesta(7).
Kehityksen plastisuus
Kehitysplastisuus ja aikuisplastisuus ovat kaksi neuroplastisuuden osa-aluetta, jotka esiintyvät eri elämänvaiheissa. Kehityksellinen plastisuus viittaa hermoston plastisuusprosessiin, joka tapahtuu aivojen kehityksen aikana alkionkehityksestä lapsuuteen ja nuoruuteen. Tänä aikana aivot ovat erittäin muokattavissa ja reagoivat kokemuksiin, ja hermoyhteydet ja -piirit muodostuvat ja tarkentuvat aistimusten ja ympäristöärsykkeiden vaikutuksesta. Kehityksellisellä plastisuudella on ratkaiseva merkitys aivojen normaalissa kehityksessä, mukaan lukien toiminnallisten hermopiirien muodostuminen ja kriittisten kognitiivisten ja käyttäytymistoimintojen luominen(8).
Aikuisten plastisuus
Sitä vastoin, aikuisten plastisuus viittaa aivojen kykyyn tehdä plastisia muutoksia vastauksena kokemuksiin tai vammoihin aikuisuudessa. Vaikka plastisuuden aste on aikuisiässä yleensä alhaisempi kuin kehityksen aikana, aikuisten aivoissa on edelleen huomattava kyky hermostolliseen plastisuuteen.
Aikuisiän plastisuus on jatkuva oppimis- ja sopeutumisprosessi, jota tapahtuu koko eliniän ajan. Sillä on ratkaiseva merkitys kognitiivisten ja käyttäytymistoimintojen ylläpitämisessä muuttuvissa ympäristöissä(9).
Yksi keskeinen ero kehityksellisen plastisuuden ja aikuisen plastisuuden välillä on plastisten muutosten luonne. Kehityksen aikana plastisuuteen liittyy usein uusien synapsien muodostumista, käyttämättömien yhteyksien karsimista sekä dendriitti- ja aksoniprosessien kasvua ja uudelleenorganisoitumista. Aikuisten plastisuuteen sen sijaan kuuluu olemassa olevien yhteyksien vahvistuminen tai heikkeneminen synaptisen voimakkuuden muutosten kautta ja uusien yhteyksien kasvu uusien synapsien muodostumisen tai uusien dendriittisten prosessien itämisen kautta.
Neuroplastisuuden keskeiset osatekijät
1. Synaptinen plastisuus
Synaptisella plastisuudella tarkoitetaan synapsien, hermosolujen välisten yhteyksien, kykyä muuttaa voimakkuuttaan toiminnan seurauksena. Se on perustavanlaatuinen mekanismi, joka on oppimisen ja muistin sekä uusien hermoyhteyksien muodostamisen taustalla. Synaptista plastisuutta voi esiintyä sekä eksitatorisissa että inhibitorisissa synapseissa, ja sitä ohjaavat muutokset välittäjäaineiden vapautumisessa ja reseptorien ilmentymisessä postsynaptisella kalvolla.
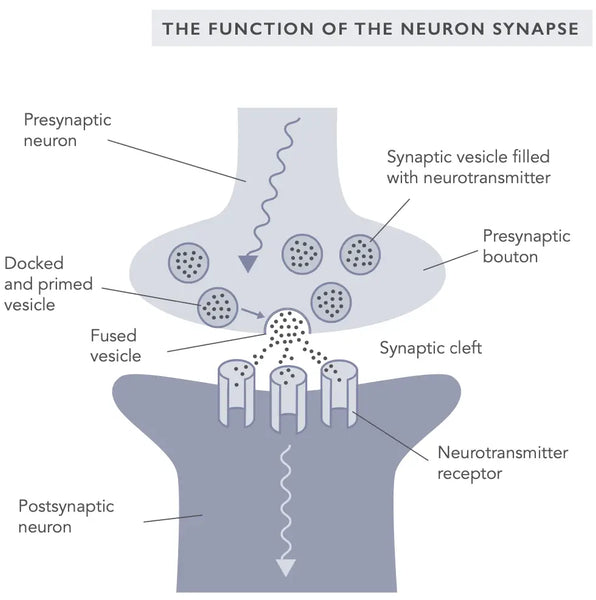
Kaksi parhaiten tutkittua synaptisen plastisuuden muotoa ovat pitkäaikainen potentiaatio (LTP) ja pitkäaikainen depressio (LTD), jotka tunnetaan myös nimellä Hebbin plastisuus (viitaten neuropsykologi Donald Hebbiin, joka esitteli synaptisen plastisuuden ensimmäisen kerran vuonna 1949).(10)
LTP on prosessi, jossa synapsien voimakkuus kasvaa toistuvan toiminnan seurauksena. Sen uskotaan olevan taustalla hermoyhteyksien vahvistumisessa oppimisen ja muistin muodostumisen aikana. LTD taas on prosessi, jossa synapsien voimakkuus vähenee matalataajuisen tai pitkäaikaisen toiminnan seurauksena. LTD:n ajatellaan vaikuttavan hermoyhteyksien heikkenemiseen unohtamisen ja sukupuuttoon kuolemisen aikana(11). Rlue lisää LTP:stä, LTD:stä ja muistinmuodostuksesta osoitteesta biohakkerin Käsikirjan Mieli -luvusta.
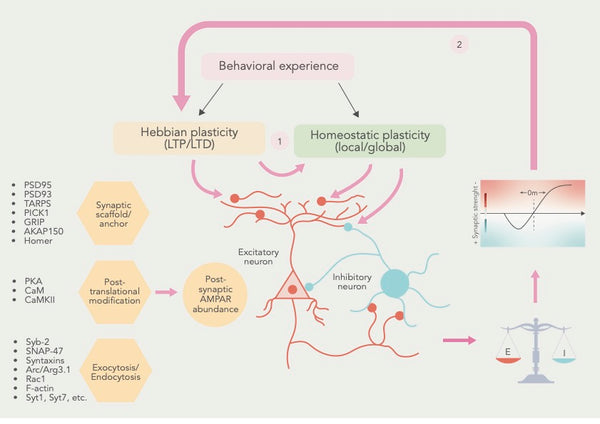
Kuva: Hebbiläisen ja homeostaattisen plastisuuden välinen yhteistyösuhde.
Lähde: Li, J. & Park, E. & Zhong, L. & Chen, L. (2019). Homeostaattinen synaptinen plastisuus metaplastisena mekanismina - molekyyli- ja solunäkökulma. Current Opinion in Neurobiology 54: 44–53.
E= synaptinen heräte I = synaptinen inhibitio
LTP:n ja LTD:n lisäksi on tunnistettu monia muitakin synaptisen plastisuuden muotoja, muun muassa seuraavat metaplastisuus, jolla tarkoitetaan muutoksia LTP:n ja LTD:n induktiokynnyksessä ja homeostaattinen plastisuus, jolla tarkoitetaan neuronien kykyä säätää aktiivisuuttaan vastauksena verkon aktiivisuuden muutoksiin.(12)
Kun nämä synaptisen plastisuuden muodot yhdistetään, tutkijat päättelevät, että Hebbin ja homeostaattinen synaptinen plastisuus yhdistyvät yhteisiin soluprosesseihin ja että homeostaattinen plastisuus säätää synapsien tilaa vaikuttaakseen Hebbin plastisuuteen (ks. kuva yllä).
Synaptista plastisuutta säätelevät erilaiset molekyyli- ja solumekanismit, kuten proteiinikinaasien ja fosfataasien aktiivisuus, proteiinien synteesi ja hajoaminen sekä geeniekspression muutokset. Nämä mekanismit reagoivat herkästi erilaisiin ympäristö- ja kokemustekijöihin, kuten aistimuksiin, stressiin ja sosiaaliseen vuorovaikutukseen(13-15).
Neurogeneesi
Neurogeneesi on prosessi, jossa aivoissa syntyy uusia hermosoluja, erityisesti aivojen hippokampuksessaalueella, joka on tärkeä oppimisen ja muistin kannalta. Se on perustavanlaatuinen mekanismi, joka on taustalla aivojen kyvyssä sopeutua ja reagoida ympäristö- ja kokemustekijöihin.
Neurogeneesi tapahtuu hippokampuksen hammaskiilteen subgranulaarisessa vyöhykkeessä, jossa hermorakenteen kantasolut synnyttävät välivaiheen esisoluja, jotka puolestaan synnyttävät epäkypsiä hermosoluja. Nämä epäkypsät neuronit siirtyvät sitten hippokampuksen granulosolukerrokseen, jossa ne kypsyvät ja integroituvat olemassa olevaan hermopiiriin. Vaikka uusia neuroneja on havaittu myös muilla alueilla, neurogeneesin laajuus näillä alueilla, kuten neokorteksissa ja hypotalamuksessa, on edelleen kiistanalainen(16).
Neurogeneesin säätely on monimutkainen ja dynaaminen prosessi, johon vaikuttavat eri tekijät, kuten genetiikka, epigenetiikka ja ympäristötekijät, kuten liikunta ja stressi. Tutkimukset ovat esimerkiksi osoittaneet, että liikunta (erityisesti aerobinen liikunta) voi stimuloida neurogeneesiä vapauttamalla kasvutekijöitä, kuten aivoista peräisin olevaa neurotrofista tekijää (BDNF) ja insuliinin kaltaista kasvutekijää-1 (IGF-1). Sitä vastoin stressin ja kroonisen tulehduksen on osoitettu heikentävän neurogeneesiä pro-inflammatorisen sytokiinin IL-1β:n välityksellä (17-19).
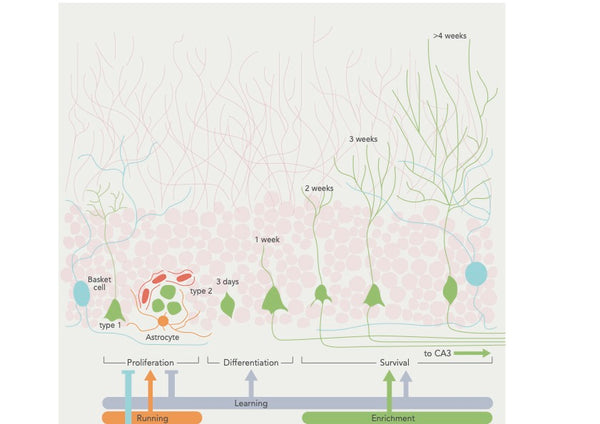
Kuva: Neurogeneesin säätely käyttäytymistekijöiden avulla.
Lähde: Aimone, J. et al. (2014). Aikuisten neurogeneesin säätely ja toiminta: geeneistä kognitioon. Fysiological Reviews 94 (4): 991–1026.
Neurogeneesin toiminnallinen rooli on edelleen aktiivisen tutkimuksen kohteena, mutta sillä uskotaan olevan merkitystä oppimisessa, muistissa, mielialan säätelyssä ja stressivasteessa. Neurogeneesin tutkimuksella on merkittäviä vaikutuksia uusien hoitomuotojen ja interventioiden kehittämiseen neurologisia ja psykiatrisia sairauksia varten(20-22).
Dendriittinen arborisaatio
Dendriittinen arborisaatio (tai dendriittinen haarautuminen) viittaa prosessiin, jonka avulla dendriititeli neuronin solurungosta lähtevät haarautuneet rakenteet, kehittyvät ja kehittävät haarautumiskuvioitaan. Tämä prosessi on elintärkeä aivojen hermopiirien kytkeytyneisyyden ja toiminnallisten ominaisuuksien muodostumiselle.
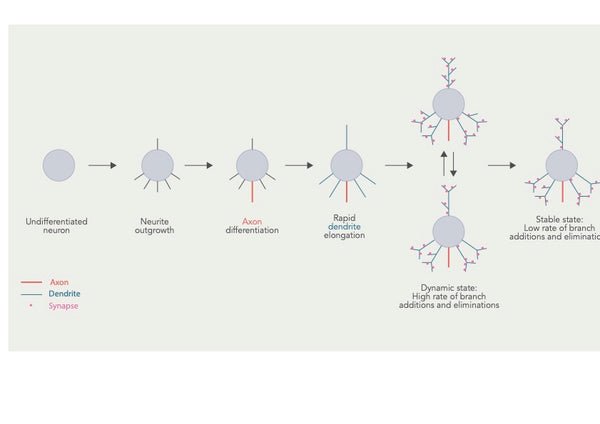 Kuva: Dendriittisen arborin kehittyminen ja useat päällekkäiset vaiheet.
Kuva: Dendriittisen arborin kehittyminen ja useat päällekkäiset vaiheet.
Lähde: Urbanska, M. & Blazejczyk, M. & Jaworski, J. (2008). Dendriittisen arborisaation molekulaarinen perusta. Acta Neurobiologiae Experimentalis 68 (2): 264–288.
Dendriittinen arborisaatio on monimutkainen prosessi, jota säätelevät eri tekijät, mukaan lukien geneettiset ja epigeneettiset tekijät sekä ympäristötekijät, kuten aistitieto ja hermoston aktiivisuus. Dendriittien kasvua ja haarautumista ohjaa solunulkoisten signaalien, kuten kasvutekijöiden ja välittäjäaineiden, aktivoimien signaalireittien aktiivisuus. Nämä signaalit voivat vaikuttaa dendriitin kasvuun ja haarautumiseen osallistuvien geenien ilmentymiseen(23).
Dendriittisen arborisaation säätely on tärkeää aivojen toiminnallisten hermopiirien muodostamisen ja ylläpitämisen kannalta. Esimerkiksi dendriittinen arborisaatio on kriittinen synapsien, hermosolujen välisten kommunikaatiopaikkojen, muodostumiselle. Dendriittien haarautumiskuvioilla voidaan vaikuttaa muodostuvien synapsien tyyppeihin ja lukumäärään, mikä voi vaikuttaa syvällisesti hermopiirien toiminnallisiin ominaisuuksiin(24).
Dendriittiarborisaatiolla on merkitystä aistitiedon käsittelyssä. Se on myös ratkaisevan tärkeä kognitiivisissa toiminnoissa ja muistin muodostumisessa. Erityisesti dendriittien haarautumiskuvioilla voidaan vaikuttaa muodostuvien synapsien tyyppeihin ja lukumäärään, mikä voi vaikuttaa syvällisesti oppimiseen ja muistiin osallistuvien hermopiirien toiminnallisiin ominaisuuksiin(25).
Tutkimukset ovat osoittaneet, että dendriittisen arborisaation muutoksia voi tapahtua vastauksena oppimiskokemuksiin, ja näiden muutosten uskotaan edistävän uusien muistojen muodostumista ja ylläpitoa. Esimerkiksi jyrsijöiden harjoittelu avaruudellisessa muistitehtävässä on lisännyt dendriittistä haarautumista hippokampuksessa, joka on avaruudellisen oppimisen ja muistin kannalta kriittinen aivoalue(26) .
Lisäksi tutkimukset ovat osoittaneet, että muutokset dendriittisessä arborisaatiossa liittyvät kognitiivisiin puutoksiin neurodegeneratiivisissa sairauksissa. Alzheimerin taudissa dendriittiset selkärangat, dendriittien rakenteet, jotka muodostavat synapseja muiden neuronien kanssa, häviävät sairastuneilla aivoalueilla, mikä johtaa heikentyneeseen synaptiseen plastisuuteen ja kognitiivisiin puutoksiin(30).
Myelinisaatio
Myelinisaatio on biologinen prosessi, jossa aksonit, neuronien pitkulaiset ja hoikat solupidennykset, jotka välittävät sähköisiä impulsseja muihin neuroneihin, päällystetään myeliiniksi kutsutulla lipidipitoisella aineella. Myeliinituppi tuottavat oligodendrosyytit keskushermostossa (CNS) ja Schwannin solut ääreishermostossa (PNS). Myeliini toimii eristeenä, jonka ansiosta sähköiset signaalit kulkevat nopeammin ja tehokkaammin aksoneja pitkin(27).
Myelinisaatioprosessi alkaa alkionkehityksen aikana ja jatkuu varhaisaikuisuuteen asti, ja eri aivojen ja hermoston alueet myelinisoituvat eri aikoina. Yleensä myelinisaatio alkaa aivorungosta ja selkäytimestä ja etenee aivokuorelle ja muille korkeammille aivoalueille(28).(28) Myeliinitupet pysyvät tyypillisesti samanpituisina pitkiä aikoja, mikä viittaa siihen, että olemassa olevan myeliinin rakenteessa ei tapahdu suuria muutoksia (ks. kuva alla)(29).
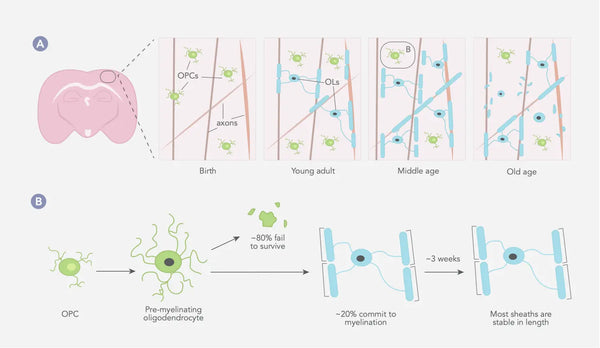
Kuva: Oligodendrosyyttien ja myeliinin dynamiikka nisäkkäiden somatosensorisessa aivokuoressa koko elämän ajan.
Lähde: Williamson, J. & Lyons, D. (2018). Myeliinin dynamiikka läpi elämän: alati muuttuva maisema? Frontiers in Cellular Neuroscience 12: 424.
OPC = Oligodendrosyyttien esiasteiden solut. OL = Oligodendrosyytit
Myelinisaation säätely on monimutkainen prosessi, johon vaikuttavat useat tekijät, kuten genetiikka, epigenetiikka ja ympäristötekijät, kuten kokemus ja hermoston toiminta. Tutkimukset ovat esimerkiksi osoittaneet, että aistikokemukset voivat vaikuttaa aivojen myelinisaation ajoitukseen ja laajuuteen. Samoin hermostollinen toiminta voi edistää myelinisaatiota vapauttamalla BDNF:n kaltaisia signaalimolekyylejä.
Keskushermostossa myelinisaatioprosessi aktivoituu aksonitoiminnan ja astrosyyttien vaikutuksesta, kun taas mikroglia/makrofagit vastaavat myeliinin poistamisesta. Kun aksonit ovat myelinisoituneet, niiden terveyden ja toimivuuden jatkuminen riippuu gliasolujen tarjoamista välttämättömistä aineenvaihduntatuotteista ja neurotrofisista tekijöistä(31).
Myelinisaation toiminnallinen rooli on ratkaisevan tärkeä hermosignaalien tehokkaan ja tuloksellisen välittämisen kannalta aivoissa ja hermostossa. Myelinisaatio on avainasemassa kognitiivisissa ja motorisissa toiminnoissa, kuten tarkkaavaisuudessa, oppimisessa ja koordinaatiossa. Lisäksi myelinisaatio on välttämätöntä myös valkean aineen kehittymiselle, joka on aivojen aksoniyhteyksien verkosto, jonka avulla eri aivoalueet voivat kommunikoida ja koordinoida toimintaansa(32).
Aivokuoren uudelleenorganisoituminen
Aivokuoren uudelleenorganisoituminen, joka tunnetaan myös nimellä aivokuoren plastisuustarkoittaa aivojen kykyä järjestää hermoverkkojaan uudelleen vastauksena aistimusten tai muunlaisten kokemusten muutoksiin. Tämä prosessi on ratkaisevan tärkeä toiminnallisten hermopiirien kehittymiselle ja aivojen kyvylle sopeutua ympäristön muutoksiin. Aivokuoren uudelleenorganisoitumista tapahtuu useilla aivojen tasoilla, primaarisista aistialueista korkeamman tason assosiaatioalueisiin(33).
Aivokuoren uudelleenorganisoitumisen taustalla oleviin mekanismeihin liittyy muutoksia synaptisen voimakkuuden ja neuronien kytkeytyneisyyden suhteen. Tutkimukset ovat esimerkiksi osoittaneet, että muutokset aistituloissa voivat johtaa muutoksiin synapsien tehossa ja määrässä aivokuoren alueilla, joihin muutokset vaikuttavat. Vastaavasti muutokset kokemuksissa tai käyttäytymisessä voivat johtaa muutoksiin neuraalisen aktiivisuuden mallissa ja synaptisten yhteyksien voimakkuudessa ja spesifisyydessä(34).
Aivokuoren uudelleenjärjestelyn toiminnalliset seuraukset voivat olla hyödyllisiä tai haitallisia. Toisaalta aivokuoren uudelleenorganisoituminen voi mahdollistaa aivojen sopeutumisen ympäristön muutoksiin ja toipumisen vammoista tai sairauksista. Toisaalta kortikaalinen uudelleenorganisoituminen voi myös edistää maladaptiivisten hermopiirien ja kroonisten kipuoireyhtymien kehittymistä.
Toinen esimerkki on tinnitus, tila, jossa yksilöt havaitsevat soinnin tai muun äänen ilman ulkoista ärsykettä. Tutkimukset ovat osoittaneet, että kuuloaivokuoren aivokuoren kortikaalinen uudelleenorganisoituminen voi vaikuttaa tinnituksen kehittymiseen ja ylläpitoon. Erityisesti aivot voivat järjestäytyä uudelleen vastauksena kuulojärjestelmän vaurioitumiseen, mikä johtaa haamuäänien havaitsemiseen.(35)
Interventiot, jotka edistävät aivokuoren uudelleenorganisoitumista (esim. aivojen plastisuuteen perustuvat terapiat), voivat olla hyödyllisiä kroonisten kipuoireyhtymien, aivohalvauksen ja muiden neurologisten vaurioiden hoidossa(36).
Neuroplastisuutta lisäävät luonnolliset tekijät
Useiden luonnollisten ja teknologisten menetelmien on osoitettu edistävän neuroplastisuutta ja parantavan aivotoimintaa.
Alla on lueteltu parhaat yleiset elämäntapatekijät, jotka parantavat neuroplastisuutta:
- Nukkuminen: Riittävä uni (ja syvä uni) on aivojen toiminnan kannalta olennainen, ja sen on osoitettu edistävän neuroplastisuutta tehostamalla synaptista plastisuutta ja helpottamalla muistojen vakiinnuttamista ja lisäämällä oppimiskykyä(37-38).(37-38).
- Liikunta: Liikunnan on osoitettu lisäävän neuroplastisuutta edistämällä uusien hermosolujen muodostumista, tehostamalla dendriittisten selkärankojen kasvua ja parantamalla olemassa olevien hermoverkkojen toimintaa. Erityisesti aerobisen liikunnan on osoitettu lisäävän neurotrofisia tekijöitä (BDNF, NGF ja GDNF ), jotka ovat proteiineja, jotka edistävät neuronien ja gliasolujen kasvua ja selviytymistä.(39-41)
- Meditaatio: Mindfulness-meditaation on osoitettu edistävän neuroplastisuutta lisäämällä harmaan aineen tiheyttä aivojen alueilla, jotka liittyvät tarkkaavaisuuteen, tunteiden säätelyyn ja itsetuntemukseen. Se voi myös parantaa valkoisen aineen eheyttä, joka on elintärkeää eri aivoalueiden väliselle viestinnälle(42-44).
- Ajoittainen paasto: Ajoittainen paasto, jossa päivittäinen ruoan saanti rajoitetaan tiettyihin tunteihin, parantaa neuroplastisuutta edistämällä uusien neuronien kasvua ja synaptista plastisuutta. Se voi myös parantaa kognitiivisia toimintoja ja vähentää hermoston rappeutumissairauksien riskiä.(45)
- Ajoittainen aineenvaihdunnan vaihtaminen (IMS).: Elämäntyylin, johon kuuluu vuorottelevia metabolisen stressin ja palautumisen jaksoja, kuten paasto ja liikunta, jota seuraa syöminen, lepo ja uni, on ehdotettu parantavan aivojen toimintaa ja kimmoisuutta. IMS voi edistää kognitiivisia kykyjä ja emotionaalista hyvinvointia tukevien hermopiirien terveyttä ja toimintaa koko elämän ajan. Se vaikuttaa laajasti useisiin signalointireitteihin, jotka lisäävät neuroplastisuutta ja parantavat aivojen kykyä kestää vammoja ja sairauksia(46).
- Sosiaalinen sitoutuminen: Sosiaalisen vuorovaikutuksen ja sitoutumisen on osoitettu edistävän neuroplastisuutta lisäämällä uusien hermosolujen kasvua ja parantamalla olemassa olevien hermoverkkojen toimintaa. Joidenkin tutkimusten mukaan sosiaalinen sitoutuminen voi jopa suojata kognitiiviselta heikkenemiseltä ja hermoston rappeutumissairauksien, kuten Alzheimerin taudin, puhkeamiselta. Siksi vahvan sosiaalisen verkoston ylläpitäminen ja säännöllinen sosiaalinen toiminta voivat olla tehokas tapa tukea ja parantaa neuroplastisuutta koko eliniän ajan.(47-48)
- Ympäristön rikastuttaminen: Lähestymistapa, jossa organismin elinolosuhteet optimoidaan siten, että ne tarjoavat monipuolista sensorista, kognitiivista ja motorista stimulaatiota. Tämän strategian on todettu edistävän neuroplastisuutta aiheuttamalla muutoksia hermoston aktiivisuudessa ja morfologiassa. Erityisesti ympäristön rikastamisen on osoitettu lisäävän uusien hermosolujen kasvua, edistävän synaptista plastisuutta ja parantavan olemassa olevien hermoverkkojen toimintaa, mikä johtaa parempiin kognitiivisiin, käyttäytymiseen liittyviin ja emotionaalisiin tuloksiin.(49-50)
- Kognitiivinen koulutus: Aivoja haastavia toimintoja, jotka parantavat aivojen neuroplastisuutta. Tällaisia ovat esimerkiksi uuden kielen oppiminen, soittimen soittaminen tai arvoitusten ratkaiseminen - ne voivat parantaa neuroplastisuutta edistämällä uusien neuronien kasvua ja synaptista plastisuutta.(51-52)
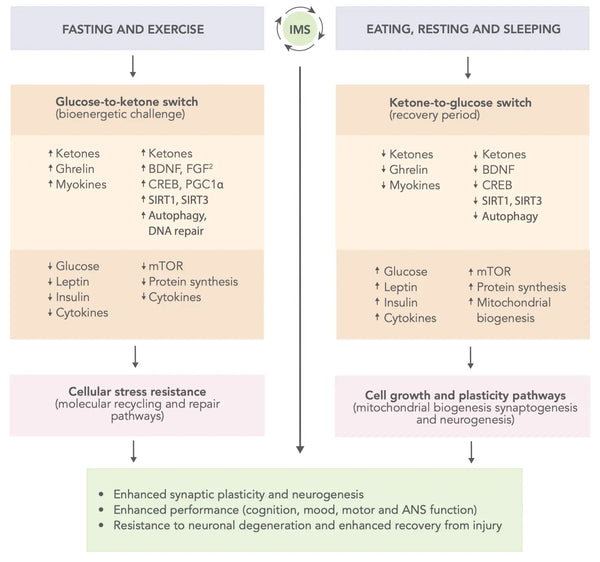
Kuva: Kaavamainen malli siitä, miten ajoittainen aineenvaihdunnan vaihtaminen voi optimoida aivojen suorituskykyä ja lisätä vastustuskykyä vammoja ja sairauksia vastaan.
Lähde: Mattson, M. & Moehl, K. & Ghena, N. & Schmaedick, M. & Cheng, A. (2018). Ajoittainen metabolinen vaihtelu, neuroplastisuus ja aivojen terveys. Nature Reviews Neuroscience 19 (2): 81–94.
Neuroplastisuutta tukevat ravitsemukselliset tekijät
- Omega-3-rasvahapot: Pitkäketjuisten omega-3-rasvahappojen, erityisesti dokosaheksaeenihapon (DHA), on osoitettu edistävän neuroplastisuutta tehostamalla synaptista plastisuutta ja lisäämällä dendriittisten selkärankojen kasvua. Ne voivat myös vähentää aivojen tulehdusta, joka voi heikentää neuroplastisuutta. Omega-3-rasvahappojen lähteitä ovat rasvaiset kalat, kuten lohi ja sardiinit, sekä ravintolisät.(53-55)
- Kurkumiini: Kurkumassa olevan kurkumiiniyhdisteen on osoitettu tehostavan neuroplastisuutta edistämällä uusien hermosolujen kasvua ja vahvistamalla synaptista plastisuutta. Sillä voi olla myös tulehdusta ehkäiseviä vaikutuksia, jotka voivat parantaa aivojen toimintaa. Mielenkiintoista on, että kurkumiini voi myös kumota kroonisen stressin aiheuttaman kognition ja hermosolujen plastisuuden heikkenemisen(56-57).(56-57) - Kokeile AGEless Defense -valmistetta, joka sisältää B-vitamiineja, polyfenoleja, aminohappoja ja kurkumiinia.
- B-vitamiinit: B-kompleksivitamiinien ja koliinin on osoitettu parantavan aivojen neuroplastisuutta. Niillä on ratkaiseva rooli aivojen toimintaa tukevissa erilaisissa aineenvaihduntareiteissä, kuten välittäjäaineiden ja myeliinin syntetisoinnissa. B-vitamiinit, erityisesti B12-vitamiini ja folaatti, osallistuvat myös DNA:n metylaatioon, joka voi vaikuttaa geeniekspressioon neuroplastisuudessa(58-62).
- D-vitamiini: Joidenkin todisteiden mukaan D-vitamiinilla voi olla merkitystä neuroplastisuuden edistämisessä. D-vitamiinireseptoreita on löydetty eri aivoalueilta, myös hippokampuksesta, joka osallistuu oppimiseen ja muistiin. Eläimillä ja ihmisillä tehdyissä tutkimuksissa on saatu viitteitä siitä, että D-vitamiinin puute voi heikentää kognitiivisia toimintoja ja vähentää neuroplastisuuden edistämisen kannalta olennaisten erityisten neurotrofisten tekijöiden tuotantoa(63-64) . Hanki Ecosh K2+D3-vitamiinia maksimoidaksesi neuroplastisuuden.
- Polyfenolit: Polyfenolien on osoitettu parantavan aivojen neuroplastisuutta. Yksi mekanismeista on niiden kyky moduloida synaptiseen plastisuuteen ja neurogeneesiin liittyviä signalointireittejä. Niillä voi olla myös tulehdusta ehkäiseviä vaikutuksia ja ne voivat suojata hapettumisstressiltä, parantaa neuronien toimintaa ja edistää neuroplastisuutta. Yleisesti ottaen polyfenolit voivat ehkäistä neurodegeneratiivisten patologioiden etenemistä. 65-66. - Kokeile yhtä parhaista polyfenoleista: Purovitalis Liposomal Quercetin.
Neuroplastisuutta tukevat teknologiset menetelmät
- Transkraniaalinen magneettistimulaatio (TMS).: TMS on ei-invasiivinen tekniikka, joka käyttää magneettikenttiä stimuloimaan hermotoimintaa tietyillä aivoalueilla. Sen on osoitettu lisäävän neuroplastisuutta ja parantavan kognitiivisia toimintoja eri yhteyksissä, muun muassa masennuksesta, ahdistuksesta ja aivohalvauksesta kärsivillä henkilöillä(67-69).(67-69) - - Hanki NeoRhythm OmniPEMF -neurostimulaatiolaite täältä. [katso kuva alla]
- Aivostimulaatio: TMS:n lisäksi muiden aivostimulaatiomuotojen, kuten transkraniaalisen tasavirtastimulaation (tDCS) ja transkraniaalisen vaihtovirtastimulaation (tACS), on osoitettu lisäävän neuroplastisuutta ja parantavan kognitiivisia toimintoja(70-71).(70-71) Lue lisää tDCS:stä biohakkerikäsikirjasta.
- Neurofeedback: Neurofeedback on tekniikka, jossa seurataan aivojen sähköistä toimintaa ja annetaan palautetta yksilölle reaaliajassa. Sen on osoitettu tehostavan neuroplastisuutta edistämällä uusien neuronien kasvua ja synaptista plastisuutta. Neurofeedbackia on käytetty tarkkaavaisuus- ja ylivilkkaushäiriön (ADHD), ahdistuneisuuden ja muiden neurologisten sairauksien hoitoon.(72-74) Lue lisää neurofeedbackista osoitteesta biohakkeri's Handbook.
- Virtuaalitodellisuus (VR): Virtuaalitodellisuus on immersiivinen teknologia, jota käytetään koulutukseen, kuntoutukseen ja terapeuttisiin tarkoituksiin. Viime vuosina yhä suurempi kiinnostus on kohdistunut VR:n käyttöön aivojen neuroplastisuuden parantamisessa. VR voi parantaa neuroplastisuutta tarjoamalla stimuloivan ja kiinnostavan ympäristön, joka haastaa aivot sopeutumaan ja oppimaan. VR voi esimerkiksi simuloida tosielämän skenaarioita ja tarjota mahdollisuuksia oppimiseen ja harjoitteluun turvallisessa ja kontrolloidussa ympäristössä. Tämä voi edistää uusien hermosolujen kasvua ja parantaa synaptista plastisuutta.(75-76)
- Kognitiiviset koulutusohjelmistot: Kognitiivisessa harjoitteluohjelmistossa käytetään tietokoneohjelmia kognitiivisten toimintojen parantamiseksi haastamalla aivot muistiharjoituksilla, ongelmanratkaisutehtävillä ja tarkkaavaisuustehtävillä. Lisäksi henkinen harjoittelu voi stimuloida tiettyjen välittäjäaineiden, kuten dopamiinin ja asetyylikoliinin, vapautumista, mikä voi parantaa synaptista plastisuutta ja kognitiivisia toimintoja sekä lisätä BDNF:n määrää aivoissa.(77-79)
Johtopäätös
Yhteenvetona voidaan todeta, että aivojen huomattava kyky muokata itseään neuroplastisuuden eri muotojen kautta - kehitys-, aikuis-, rakenne- ja toimintakyky - avaa monia mahdollisuuksia henkilökohtaiseen kasvuun ja kognitiiviseen parantamiseen. Tämän artikkelin strategioiden omaksuminen antaa sinulle työkalut, joilla voit lähteä transformatiiviselle matkalle. Muokkaat aktiivisesti aivojesi tulevaisuutta tekemällä mielen harjoituksia, vaalimalla tietoisuutta ja asettamalla terveelliset tavat etusijalle. Muista, että avain on johdonmukaisuudessa ja omistautumisessa. Tartu siis tähän jännittävään seikkailuun, jossa avaudut aivojesi potentiaalille.
Jos haluat viedä pelisi seuraavalle oktaaville, ennakkotilaa massiivinen jatko-osamme osoitteesta biohakkerin Käsikirjaa, joka on Resilientti oleminen -kirja.
P.S. Tämä artikkeli perustuu Resilientti oleminen -kirjan Henkinen kestävyys -osion tekstiin.
Lähdeviitteet:
- Puderbaugh, M. & Emmady, P. (2022). Neuroplastisuus. In . StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing.
- Venkataramani, P. (2010). Santiago Ramón y Cajal: neurotieteiden isä. Resonanssi 15 (11): 968–976.
- Draganski, B. & Gaser, C. & Busch, V. & Schuierer, G. & Bogdahn, U. & May, A. (2004). Harjoittelun aiheuttamat muutokset harmaassa aineessa. Nature 427 (6972): 311–312.
- Kleim, J. & Jones, T. (2008). Kokemuksesta riippuvaisen neuraalisen plastisuuden periaatteet: vaikutukset aivovaurion jälkeiseen kuntoutukseen. Journal of Speech Language and Hearing Research 51: S225-S239.
- Bozelos, P. & Poirazi, P. (2017). Rakenteellisen plastisuuden vaikutus muistin kapasiteettiin. In . The Rewiring Brain (s. 319-341). Cambridge (USA): Academic Press.
- Grafman, J. (2000). Toiminnallisen neuroplastisuuden käsitteellistäminen. Journal of Communication Disorders 33 (4): 345–356.
- Taubert, M. et al. (2010). Ihmisen aivorakenteen dynaamiset ominaisuudet: oppimiseen liittyvät muutokset aivokuorialueilla ja niihin liittyvissä kuituyhteyksissä. Journal of Neuroscience 30 (35): 11670–11677.
- Kolb, B. & Gibb, R. (2011). Aivojen plastisuus ja käyttäytyminen kehittyvissä aivoissa. Journal of the Canadian Academy of Child and Adolescent Psychiatry. 20 (4): 265–276.
- Fuchs, E. & Flügge, G. (2014). Aikuisten neuroplastisuus: yli 40 vuotta tutkimusta. Neural Plasticity 2014: 541870
- Hebb, D. (1949). Käyttäytymisen organisointi: Neuropsykologinen teoria. New York: Wiley and Sons.
- Bliss, T. & Collingridge, G. (1993). Muistin synaptinen malli: pitkäaikainen potentiaatio hippokampuksessa. Nature 361 (6407): 31–39.
- Li, J. & Park, E. & Zhong, L. & Chen, L. (2019). Homeostaattinen synaptinen plastisuus metaplastisena mekanismina - molekyyli- ja solunäkökulma. Current Opinion in Neurobiology 54: 44–53.
- Magee, J. & Grienberger, C. (2020). Synaptisen plastisuuden muodot ja toiminnot. Annual Review of Neuroscience 43: 95–117.
- Vitureira, N. & De Pasquale, R. & Leão, R. & Rossi, F. (2022). Synaptisen plastisuuden solu- ja molekyylimekanismit hippokampuksen ja aivokuoren synapseissa. Frontiers in Cellular Neuroscience 16: 980623.
- Fox, K. & Stryker, M. (2017). Hebbiläisen ja homeostaattisen plastisuuden integrointi: johdanto. Philosophical Transactions of the Royal Society B: Biological Sciences. 372 (1715): 20160413.
- Aimone, J. et al. (2014). Aikuisten neurogeneesin säätely ja toiminta: geeneistä kognitioon. Fysiological Reviews 94 (4): 991–1026
- Cotman, C. & Berchtold, N. (2002). Liikunta: käyttäytymiseen perustuva interventio aivojen terveyden ja plastisuuden parantamiseksi. Trends in Neurosciences 25 (6): 295–301.
- Vecchio, L. et al. (2018). Liikunnan neuroprotektiiviset vaikutukset: terveiden aivojen ylläpitäminen koko ikääntymisen ajan. Brain Plasticity 4 (1): 17–52.
-
Saxe, M. ym. (2006). Hippokampuksen neurogeneesin ablaatio heikentää kontekstuaalista pelon ehdollistamista ja synaptista plastisuutta dentate gyrusissa. Proceedings of the National Academy of Sciences 103 (46): 17501–17506.
- Aimone, J. et al. (2014). Aikuisten neurogeneesin säätely ja toiminta: geeneistä kognitioon. Fysiological Reviews 94 (4): 991–1026.
- Ming, G. & Song, H. (2011). Aikuisten neurogeneesi nisäkkäiden aivoissa: merkittäviä vastauksia ja merkittäviä kysymyksiä. Neuron 70 (4): 687–702.
- Urbanska, M. & Blazejczyk, M. & Jaworski, J. (2008). Dendriittisen arborisaation molekulaarinen perusta. Acta Neurobiologiae Experimentalis 68 (2): 264–288.
- Cline, H. (2001). Dendriittisen arborin kehitys ja synaptogeneesi. Current Opinion In Neurobiology 11 (1): 118–126.
- Kasai, H. & Fukuda, M. & Watanabe, S. & Hayashi-Takagi, A. & Noguchi, J. (2010). Dendriittisten selkärankojen rakenteellinen dynamiikka muistissa ja kognitiossa. Trends in Neurosciences 33 (3): 121–129.
- Holtmaat, A. & Svoboda, K. (2009). Kokemuksesta riippuva rakenteellinen synaptinen plastisuus nisäkkäiden aivoissa. Nature Reviews Neuroscience 10 (9): 647–658.
- Demerens, C. et al. (1996). Myelinisaation indusoiminen keskushermostossa sähköisen toiminnan avulla. Proceedings of the National Academy of Sciences 93 (18): 9887–9892.
- Harry, G. & Toews, A. (1998). Myelinisaatio, dysmyelinisaatio ja demyelinisaatio. Kehityksellisen neurotoksikologian käsikirja. 87–115.
- Williamson, J. & Lyons, D. (2018). Myeliinin dynamiikka läpi elämän: alati muuttuva maisema? Frontiers in Cellular Neuroscience 12: 424.
- Scheff, S. & Price, D. & Schmitt, F. & Mufson, E. (2006). Hippokampuksen synaptinen menetys varhaisessa Alzheimerin taudissa ja lievässä kognitiivisessa heikentymisessä. Neurobiology of Aging 27 (10): 1372–1384.
- Nave, K. & Werner, H. (2014). Hermoston myelinisaatio: mekanismit ja toiminnot. Annual Review of Cell and Developmental Biology 30: 503–533
- Fields, R. (2015). Uusi hermoston plastisuuden mekanismi: aktiivisuudesta riippuvainen myelinisaatio. Nature Reviews Neuroscience 16 (12): 756–767.
- Pascual-Leone, A. & Amedi, A. & Fregni, F. & Merabet, L. (2005). Ihmisen plastinen aivokuori. Annual Reviews Neuroscience 28: 377–401.
- Schoups, A. & Vogels, R. & Qian, N. & Orban, G. (2001). Suuntautumisen tunnistamisen harjoittelu parantaa suuntautumisen koodausta V1-neuroneissa. Nature 412 (6846): 549–553.
- Mühlnickel, W. & Elbert, T. & Taub, E. & Flor, H. (1998). Kuuloaivokuoren uudelleenorganisoituminen tinnituksessa. Proceedings of the National Academy of Sciences 95 (17): 10340–10343.
- Merzenich, M. & Van Vleet, T. & Nahum, M. (2014). Aivojen plastisuuteen perustuvat terapiat. Frontiers in Human Neuroscience 8: 385.
- Fattinger, S. et al. (2017). Syvä uni ylläpitää ihmisen aivojen oppimistehokkuutta. Nature Communications 8 (1): 15405.
- Voss, M. et al. (2010). Aivoverkkojen plastisuus ikääntyneiden aikuisten liikuntaharjoittelun satunnaistetussa interventiotutkimuksessa. Frontiers in Aging Neuroscience 2: 32.
- e Sousa Fernandes, M. et al. (2020). Liikunnan vaikutukset neuroplastisuuteen ja aivotoimintaan: systemaattinen katsaus ihmis- ja eläinkokeisiin. Neural Plasticity 2020: 8856621
- Hölzel, B. et al. (2011). Mindfulness-harjoittelu johtaa aivojen alueellisen harmaan aineen tiheyden lisääntymiseen. Psykiatrian tutkimus: Neuroimaging 191 (1): 36–43.
- Tang, Y. & Hölzel, B. K. & Posner, M. (2015). Mindfulness-meditaation neurotiede. Nature Reviews Neuroscience 16 (4): 213–225.
- Lardone, A. et al. (2018). Mindfulness-meditaatio liittyy hippokampuksen toiminnallisen topologian pitkäkestoisiin muutoksiin lepotilan aikana: magnetoenkefalografiatutkimus. Neural Plasticity 2018: 5340717.
- Brocchi, A. & Rebelos, E. & Dardano, A. & Mantuano, M. & Daniele, G. (2022). Ajoittaisen paaston vaikutukset aivojen aineenvaihduntaan. Nutrients 14 (6): 1275.
- Mattson, M. & Moehl, K. & Ghena, N. & Schmaedick, M. & Cheng, A. (2018). Ajoittainen aineenvaihduntakytkentä, neuroplastisuus ja aivojen terveys. Nature Reviews Neuroscience 19 (2): 81–94.
- Kramer, A. & Bherer, L. & Colcombe, S. & Dong, W. & Greenough, W. (2004). Ympäristön vaikutukset kognitiiviseen ja aivojen plastisuuteen ikääntymisen aikana. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences (Gerontologian aikakauslehdet, sarja A: Biologiset tieteet ja lääketieteelliset tieteet). 59 (9): M940-M957.
- Fratiglioni, L. & Paillard-Borg, S. & Winblad, B. (2004). Aktiivinen ja sosiaalisesti integroitunut elämäntapa myöhäisessä iässä saattaa suojata dementialta. The Lancet Neurology 3 (6): 343–353.
- Kempermann, G. (2015). Aktiivisuusriippuvuus ja ikääntyminen aikuisten neurogeneesin säätelyssä. Cold Spring Harbor Perspectives in Biology 7 (11): a018929.
- Van Praag, H. & Kempermann, G. & Gage, F. (2000). Neuraaliset seuraukset ympäristön rikastumisesta. Nature Reviews Neuroscience 1 (3): 191–198
- Lövdén, M. & Bäckman, L. & Lindenberger, U. & Schaefer, S. & Schmiedek, F. (2010). Teoreettinen kehys aikuisten kognitiivisen plastisuuden tutkimiselle. Psychological Bulletin 136 (4): 659–676
- Park, D. & Bischof, G. (2013). Ikääntyvä mieli: neuroplastisuus vastauksena kognitiiviseen harjoitteluun. Dialogues in Clinical Neuroscience 15 (1): 109–119.
- Crupi, R. & Marino, A. & Cuzzocrea, S. (2013). n-3-rasvahapot: rooli neurogeneesissä ja neuroplastisuudessa. Current Medicinal Chemistry 20 (24): 2953–2963.
- Swanson, D. & Block, R. & Mousa, S. A. (2012). Omega-3-rasvahapot EPA ja DHA: terveyshyödyt läpi elämän. Advances in Nutrition 3 (1): 1–7.
- Dyall, S. (2015). Pitkäketjuiset omega-3-rasvahapot ja aivot: katsaus EPA:n, DPA:n ja DHA:n itsenäisiin ja yhteisiin vaikutuksiin. Frontiers in Aging Neuroscience 7: 52.
- Maharjan, R. et al. (2020). Elämäntapojen rooli neuroplastisuudessa ja neurogeneesissä ikääntyvissä aivoissa. Cureus 12 (9): e10639.
- Xu, Y. et al. (2009). Kurkumiini kumoaa kroonisen stressin aiheuttaman heikentyneen kognition ja neuronaalisen plastisuuden. Neuropharmacology 57 (4): 463–471.
- Echeverry, M. et al. (2021). D- ja B 12 -vitamiinit, muuttunut synaptinen plastisuus ja ekstrasellulaarinen matriisi. In B-kompleksivitamiinit-lähteet, saanti ja uudet sovellukset.. IntechOpen.
- Downey, L. et al. (2019). Lisääntynyt posteriorinen cingulaarinen toiminnallinen liitettävyys 6 kuukauden korkean annoksen B-vitamiinien monivitamiinilisän jälkeen: satunnaistettu, kaksoissokkoutettu, lumekontrolloitu tutkimus. Frontiers in Nutrition 6: 156.
- Mattson, M. & Shea, T. (2003). Folaatti- ja homokysteiinimetabolia hermoston plastisuudessa ja hermoston rappeutumishäiriöissä. Trends in Neurosciences 26 (3): 137–146.
- Chin, E. & Goh, E. (2019). Neuronaalisen plastisuuden modulointi koliinilla. Neural Regeneration Research 14 (10): 1697.
- Jadavji, N. & Emmerson, J. & MacFarlane, A. & Willmore, W. & Smith, P. (2017). B-vitamiinien ja koliinin lisäys lisää neuroplastisuutta ja toipumista aivohalvauksen jälkeen. Neurobiology of Disease 103: 89–100.
- Mayne, P. & Burne, T. (2019). D-vitamiini synaptisessa plastisuudessa, kognitiivisessa toiminnassa ja neuropsykiatrisissa sairauksissa. Trends in Neurosciences 42 (4): 293–306.
- Echeverry, M. et al. (2021). D- ja B 12 -vitamiinit, muuttunut synaptinen plastisuus ja ekstrasellulaarinen matriisi. In B-kompleksivitamiinit-lähteet, saanti ja uudet sovellukset.. IntechOpen.
- Vauzour, D. (2012). Ruokavalion polyfenolit aivotoimintojen modulaattoreina: biologiset toimet ja molekyylimekanismit, jotka tukevat niiden suotuisia vaikutuksia. Oksidatiivinen lääketiede ja solujen pitkäikäisyys. 2012: 914273.
- Figueira, I. & Menezes, R. & Macedo, D. & Costa, I. & Nunes dos Santos, C. (2017). Polyfenolit esteiden takana: vilkaisu aivoihin. Current Neuropharmacology 15 (4): 562–594.
- Hallett, M. (2007). Transkraniaalinen magneettistimulaatio: alustus. Neuron 55 (2): 187–199.
- Jannati, A. & Oberman, L. & Rotenberg, A. & Pascual-Leone, A. (2023). Aivojen plastisuuden mekanismien arviointi transkraniaalisella magneettistimulaatiolla. Neuropsykofarmakologia 48 (1): 191–208.
- Auriat, A. & Neva, J. & Peters, S. & Ferris, J. & Boyd, L. (2015). Katsaus transkraniaaliseen magneettistimulaatioon ja multimodaaliseen neurokuvantamiseen aivohalvauksen jälkeisen neuroplastisuuden kuvaamiseksi. Frontiers in Neurology 6: 226.
- Kricheldorff, J. et al. (2022). Todisteet neuroplastisista muutoksista transkraniaalisen magneetti-, sähkö- ja syväaivostimulaation jälkeen. Brain Sciences 12 (7): 929.
- Brunoni, A. et al. (2012). Kliininen tutkimus transkraniaalisella tasavirtastimulaatiolla (tDCS): haasteet ja tulevaisuuden suunnat. Brain Stimulation 5 (3): 175–195.
- Gruzelier, J. (2014). EEG-neurofeedback suorituskyvyn optimoimiseksi. III: katsaus metodologisiin ja teoreettisiin näkökohtiin. Neuroscience & Biobehavioral Reviews. 44: 159–182.
- Trambaiolli, L. & Cassani, R. & Mehler, D. & Falk, T. (2021). Neurofeedback ja ikääntyvät aivot: systemaattinen katsaus dementiaa ja lievää kognitiivista heikkenemistä koskeviin harjoitusprotokolliin. Frontiers in Aging Neuroscience 13: 682683.
- Sitaram, R. et al. (2017). Suljetun silmukan aivokoulutus: neurofeedbackin tiede. Nature Reviews Neuroscience 18 (2): 86–100.
- Laver, K. & George, S. & Thomas, S. & Deutsch, J. & Crotty, M. (2015). Virtuaalitodellisuus aivohalvauksen kuntoutuksessa: Cochrane-katsauksen lyhennetty versio. European Journal of Physical and Rehabilitation Medicine 51 (4): 497–506.
- Huang, C. et al. (2022). Virtuaalitodellisuuteen perustuvan motorisen kontrollin harjoittelun vaikutukset tulehdukseen, oksidatiiviseen stressiin, neuroplastisuuteen ja yläraajan motoriseen toimintaan kroonista aivohalvausta sairastavilla potilailla: satunnaistettu kontrolloitu tutkimus. BMC Neurology 22 (1): 21.
- Anguera, J. et al. (2013). Videopeliharjoittelu parantaa kognitiivista kontrollia iäkkäillä aikuisilla. Nature 501 (7465): 97–101.
- Lampit, A. et al. (2014). Valvotun tietokoneavusteisen kognitiivisen harjoittelun avulla saavutettujen kognitiivisten saavutusten ajallinen kehitys: satunnaistettu, aktiivisesti kontrolloitu tutkimus vanhuksilla, joilla on useita dementian riskitekijöitä. The Journal of Prevention of Alzheimers Disease 1 (1): 33–39.
- D'Antonio, J. et al. (2019). Kognitiivinen harjoittelu ja neuroplastisuus lievässä kognitiivisessa heikentymisessä (COG-IT): kahden toimipaikan sokkoutetun, satunnaistetun, kontrolloidun hoitotutkimuksen protokolla. BMJ Open 9 (8): e028536.







