In a recent study published in the prestigious scientific journal Cell, López-Otín and colleagues (2023) presented an updated version of their widely acclaimed theory on the "hallmarks of aging." This theory proposes that the aging process is driven by biological mechanisms or "hallmarks" that contribute to the decline of cellular and tissue functions over time. This comprehensive summary article will delve into the details of this groundbreaking study and its implications for our understanding of the aging process.
Introduction
Aging is a complex and multifactorial process that affects all living organisms, from single-celled organisms to multicellular organisms like humans. Despite extensive research efforts over the years, the underlying causes of aging still need to be better understood. In 2013, López-Otín and colleagues proposed a new theory on the hallmarks of aging, which has since gained widespread recognition and influenced research in the field of aging biology. In their latest study, the authors present an updated version of this theory, which considers recent advances in our understanding of the molecular mechanisms underlying aging.
The Original 9 Hallmarks of Aging
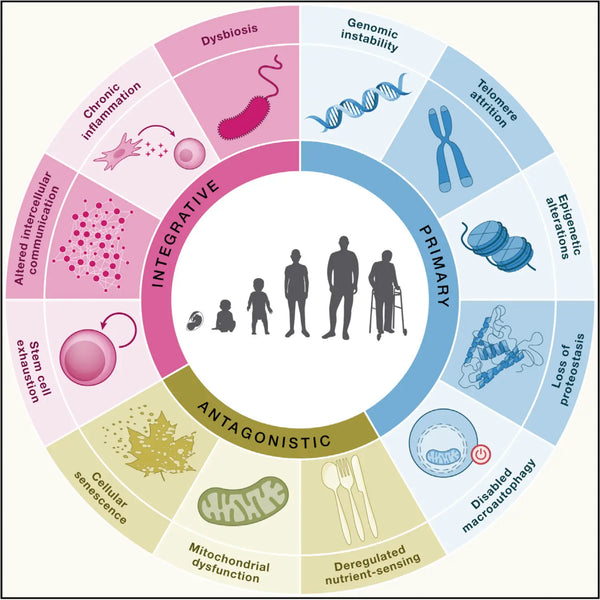
According to their original study from 2013, the hallmarks of aging theory propose that nine interconnected biological mechanisms contribute to aging. These mechanisms are:
Genomic instability
Genomic instability refers to the accumulation of DNA damage and mutations over time, which can lead to cellular dysfunction and death. This hallmark is linked to various age-related diseases, including cancer.
Telomere attrition
Telomeres are protective caps on the ends of chromosomes that shorten with each cell division. Telomere attrition can lead to cellular senescence, irreversible growth arrest, and cell death.
Epigenetic alterations
Epigenetic changes refer to modifications in gene expression patterns that do not involve changes in the underlying DNA sequence. These changes can affect various cellular processes and contribute to aging-related phenotypes.
Loss of proteostasis
Proteostasis refers to maintaining proper protein folding, trafficking, and degradation within cells. As we age, the ability of cells to maintain proteostasis declines, leading to the accumulation of misfolded and damaged proteins.
Deregulated nutrient sensing
Nutrient sensing pathways, such as the insulin/IGF-1 and mTOR pathways, regulate metabolism and growth. Dysregulation of these pathways can contribute to aging-related phenotypes, including insulin resistance and sarcopenia.
Mitochondrial dysfunction
Mitochondria are organelles that play crucial roles in energy production and cellular metabolism. Mitochondrial dysfunction can lead to oxidative stress, inflammation, and impaired cellular functions.
Cellular senescence
As mentioned earlier, cellular senescence refers to a state of irreversible growth arrest that various stressors, including DNA damage and telomere attrition, can trigger. Senescent cells can contribute to tissue dysfunction and inflammation.
Stem cell exhaustion
Stem cells are essential for tissue regeneration and repair. With age, the number and function of stem cells decline, leading to impaired tissue repair and an increased risk of age-related diseases.
Altered intercellular communication
Cell-to-cell communication is critical for the proper functioning of tissues and organs. As we age, the quality and quantity of intercellular communication decline, contributing to tissue dysfunction and disease.
What is new in their updated study, accumulating science from the past ten years?
In their latest study, "Hallmarks of Aging: An Expanding Universe," published in Cell in 2023, López-Otín and colleagues added three new hallmarks to their list: dysbiosis, chronic inflammation, and disabled macroautophagy.
Dysbiosis refers to an imbalance in the microbial community in the gut, which can lead to chronic inflammation and other adverse health outcomes. The authors propose that dysbiosis is a hallmark of aging because it is common in older adults and contributes to age-related diseases such as obesity, type 2 diabetes, and cardiovascular disease.
Chronic inflammation is another newly added hallmark of aging. Chronic inflammation occurs when the immune system is constantly activated and can lead to tissue damage and dysfunction. The authors propose that chronic inflammation is a key feature of aging and contributes to many age-related diseases, including Alzheimer's, cancer, and cardiovascular disease.
Disabled macroautophagy is the third newly added hallmark of aging. Macroautophagy is a cellular process that clears damaged or dysfunctional organelles and proteins. When macroautophagy is disabled, these damaged components can accumulate in cells, leading to cellular dysfunction and disease. The authors propose that disabled macroautophagy is a hallmark of aging because it is common in older adults and contributes to age-related diseases such as Alzheimer's disease, Parkinson's disease, and cancer.
Conclusion
López-Otín and colleagues' latest study highlights the importance of dysbiosis, chronic inflammation, and disabled macroautophagy in aging. These newly added hallmarks have been carefully studied, and their importance in developing diseases and, on the other hand, preventing them have been known for years. Therefore, it is great to see the authors expand and update their concept on the hallmarks of aging. By understanding the underlying biological mechanisms, we can develop interventions and treatments to improve healthspan and reduce the burden of age-related diseases.
---
Meanwhile, you can enjoy the carefully selected supplements for improving your healthspan in the Biohacker Center Store!
P.S. also, read the previous article (posted 09/2022) on the 9+1 hallmarks of aging and how to reverse them to get deeper into the prevention side!




